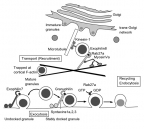
Exocytosis of docked and undocked insulin granules by different Rab27a effectors
We previously demonstrated that granuphilin, an effector of the small GTPase Rab27a, mediates the stable attachment (docking) of insulin granules to the plasma membrane. However, recent total internal reflection fluorescence (TIRF) microscopic recordings in living cells have demonstrated that granules without prior stable docking to the plasma membrane can efficiently undergo exocytosis, although the molecular mechanism is unknown. The present report is the first to identify exophilin7, another Rab27a effector expressed in pancreatic beta cell as a molecule that functions in the exocytosis of undocked granules. These findings indicate that the two Rab27a effectors, granuphilin and exophilin7, differentially regulate the exocytosis of either stably or minimally docked granules, respectively (Wang H et al., Mol. Biol. Cell, 24, 319-330, 2013).
2013/04/20